The FOCUS groundwater guidance (FOCUS 2014, p. 41) states that a reliable measured
log Kow for neutral pH must be available in order to apply the Briggs
equation. It is not clarified when it can be regarded reliable, but the
equation is stated to be produced for non-ionic compounds, suggesting that
the compound should not be ionogenic (weak acid/base)
or ionic.
Usage
TSCF(log_Kow, method = c("briggs82", "dettenmaier09"))
Arguments
- log_Kow
The decadic logarithm of the octanol-water partition constant
- method
Short name of the estimation method.
Details
The Dettenmaier equation is given to show that other views on the subject exist.
References
FOCUS (2014) Generic Guidance for Tier 1 FOCUS Ground Water Assessments.
Version 2.2, May 2014
Dettenmaier EM, Doucette WJ and Bugbee B (2009) Chemical hydrophobicity and uptake
by plant roots. Environ. Sci. Technol 43, 324 - 329
Examples
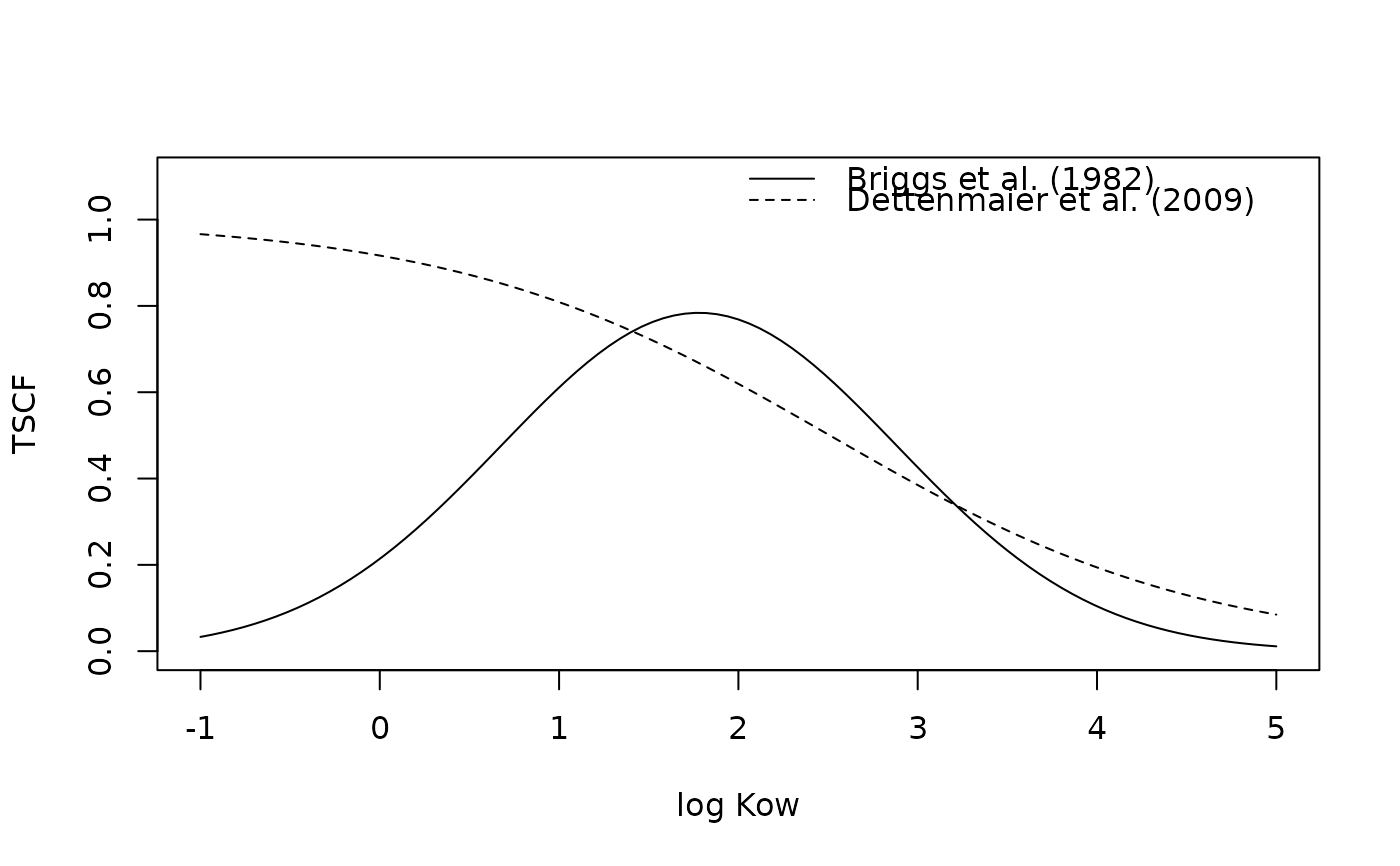
plot(TSCF, -1, 5, xlab = "log Kow", ylab = "TSCF", ylim = c(0, 1.1))
TSCF_2 <- function(x) TSCF(x, method = "dettenmaier09")
curve(TSCF_2, -1, 5, add = TRUE, lty = 2)
legend("topright", lty = 1:2, bty = "n",
legend = c("Briggs et al. (1982)", "Dettenmaier et al. (2009)"))